Description
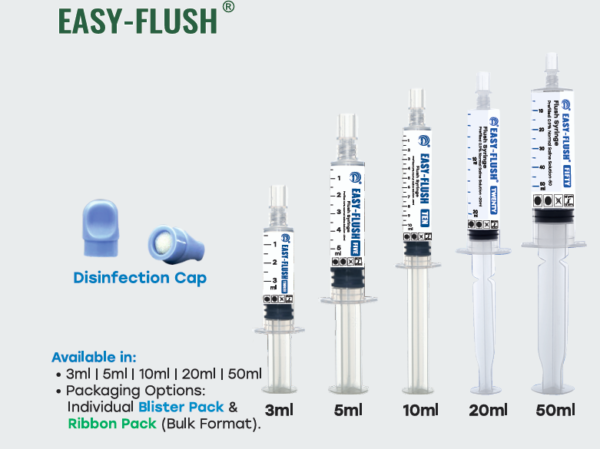
Available in
3ml, 5ml, 10ml, 20ml, & 50ml Individual Ribbon pack
Please Download eIFU – Click Here
For Sterile Variant Only
Follow manufacturer’s instructions for use and institutional procedures for Flush Syringe (Prefilled 0.9% Normal Saline Solution) With Disinfectant Cap
DESCRIPTION
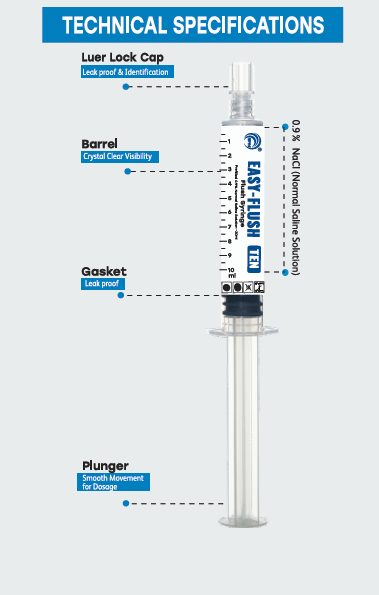
The Flush Syringe (Prefilled 0.9 % saline solution) is manufactured by SPM Medicare Pvt. Ltd. is a three-piece syringe. The syringe consists of a barrel, plunger, gasket and tip cap and it is prefilled with 0.9% normal saline solution. The Flush Syringe (Prefilled 0.9 % saline solution) comes with luer-lock connection at the tip and compatible with ISO compliant luer components for intravenous applications. The flush syringe (Prefilled 0.9% normal saline solution) is a sterile, single-use, non-toxic and non-pyrogenic medical device. The device is packed in ribbon/ Blister or paper pouch and sterilized by gamma irradiation.
Each pack of the Flush Syringe (Prefilled 0.9% Saline Solution) is supplied with a disinfectant cap as an accessory. The cap contains a PU sponge impregnated with 70% Isopropyl Alcohol (IPA) and is sealed with aluminium foil. It is designed to disinfect needle-free connectors and IV catheter hubs before access or flushing. This combination reduces the risk of infection and improves patient safety.
INTENDED PURPOSE
Flush Syringe is intended for flushing compatible intravenous administration sets and indwelling intravenous access devices.
INDICATIONS FOR USE
• Flush Syringe is designed to be used with ISO compliant luer components for intravenous applications.
• Flush Syringe is intended to be used FOR FLUSHING ONLY.
• Flush Syringe is not intended for dry product reconstitution, for medication dilution, or where intravenous therapy with sodium chloride is indicated.
INTENDED PATIENT POPULATION
Flush Syringe is intended to be used in patients irrespective of age and gender.
INTENDED USER
Flush Syringe is intended to be used by trained healthcare professionals including doctors, registered practitioners, paramedics or nursing staff experienced in the use of vascular access devices.
INTENDED USE ENVIRONMENT
Flush Syringe is intended to be used in hospitals and clinics.
CLINICAL BENEFITS
• Reduces number of steps for the user as compared to a conventional IV injection used for flushing to manually prepare a flush syringe.
• Saves time because of its ready-to-use application
• Reduces the risk of contamination due to less exposure to environment before use
PRINCIPLE OF OPERATION
Flush Syringe is used to flush out compatible intravenous administration sets and indwelling intravenous access devices. It helps to prevent blockage and remove any medicine left in the catheter. The mechanical pressure is generated in syringe through manual push to eject 0.9% w/v sodium chloride (saline) solution.
Sodium chloride is an electrolyte that is used to maintain the balance of fluid in and around the body cells and tissues once administered.
The Disinfectant Cap, used prior to flushing, contains 70% isopropyl alcohol and is placed on the needleless access device to disinfect the connector surface and provide a physical barrier against contamination, ensuring the infusion line remains sterile during use.
PERFORMANCE CHARACTERISTICS
• Interoperability with Vascular Access Devices (VAD).
• Successful Flushing
MATERIALS USED
• Barrel, Plunger & Tip Cap – Polypropylene
• Gasket – Bromo butyl rubber / TPE (Thermoplastic Elastomer) / Natural Rubber
• Gasket Mixing – Silicon 1000 CST
• Barrel spray – Silicon 1000 CST
• Sodium Chloride – 0.9% w/v sodium chloride (saline) solution
• Cap – PP/PE
• Sponge – PU
• Disinfectant – 70% IPA
• Cover – Aluminium foil
CONTRAINDICATIONS
• Do not use if the patient has a known allergy to any of its components, materials, or 0.9% w/v sodium chloride solution. It may lead to an allergic response resulting in anaphylaxis.
• Do not use if the intravenous administration sets and/or indwelling intravenous access devices are contaminated by microorganisms or the human body (skin) around the access device is inflamed
• Do not use in patients suffering from hypernatremia and fluid retention when the administration of sodium or chloride could be clinically detrimental.
POTENTIAL COMPLICATIONS
• Possible complications and/or adverse reactions associated with flushing may include sepsis or septicaemia, infections (localized/systemic), exposure to bloodborne pathogens, air embolism, particulate embolism, blood clots, thrombophlebitis, leakage that may lead to hazardous drug/fluid exposure, irritation, a transitory taste or odour during flushing. The use of contaminated normal saline products may lead to infection and possibly death.
• Not using an aseptic technique and failure to adhere to flushing guidelines may lead to catheter failure, catheter related complications such as occlusion, infiltration, extravasation, erythema, swelling, pain, injury, infection, sepsis, or death.
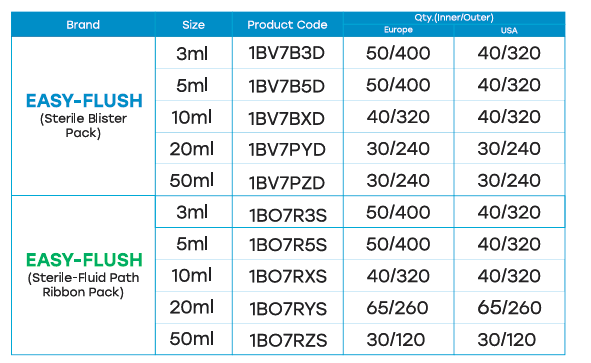
HOW SUPPLIED
Flush Syringe is supplied sterile and packed in a paper pouch. Thirty (40) such pouches are enclosed within a printed duplex box. The overall packing contains eight (8) duplex boxes and external identification stickers.
WARNINGS
• Do not reuse. Re-use may lead to infection or other illness.
• Do not re-sterilize the device by any method. Do not reprocess. It is not intended to be reprocessed and used again, even on the same patient.
INSTRUCTIONS FOR USE
• Reprocessing may compromise sterility, biocompatibility, and functional integrity of the device.
• SPM Medicare assumes no liability for the devices that have been re-use or re-sterilized.
• Do not use it if the product has been left at freezing temperature.
• Do not use the device after expiry date or the unit package or content is damaged.
• Store at specified temperature and humidity.
• Small parts are a potential choking hazard. After use, discard small parts according to the hospital protocol.
• Do not use it if the solution is cloudy or colored, contains a precipitate, or has any type of suspended particulate matter.
PRECAUTIONS
• Flush Syringe is designed to be used with ISO luer compliant components for intravenous applications
• Check the product for visual defects/cracks.
• Gradation is provided on the syringe to inject the required amount of saline.
• Avoid crushing and crimping damage to the product due to the application of surgical instruments such as forceps.
• Store at a controlled temperature (10–40°C) and humidity (55%–75%). Do not leave at freezing temperature.
• Check with drug manufacturer instructions for use to ensure compatibility with 0.9% w/v sodium chloride solution before use. If 0.9% w/v sodium chloride solution is not compatible, follow the drug manufacturer’s instructions for flushing practices, or first flush the vascular access device (VAD) with a compatible solution to remove traces of the medication in accordance with manufacturer and institution policies.
• Clinicians should consider the patient’s specific medical conditions, treatment needs, age, and weight that may require restricted sodium or fluid intake when deciding to flush with 0.9% w/v sodium chloride injection.
• Saline flushes should be taken into account when prescribing fluids to not exceed fluid intake guidelines. Follow instructions for use, and institution procedures for flush administration.
CAUTIONS:
• Carefully read all instructions prior to use.
• Do not use if the package is open or damaged.
• For single use only. Discard any partially used product.
• The solution and fluid path are sterile and non-pyrogenic if the tip cap is in place,
• syringe is intact, and there is no evidence of leakage. Use proper aseptic technique.
STORAGE
Store at a controlled temperature (10–40°C). Do not store at freezing temperature.
EXPIRATION PERIOD
The device has a shelf life of 3 years from the date of manufacturing. The product should not be used after the indicated sterility expiration date.
RETURN OF DAMAGED PRODUCT
Return the product in its original packing identified by the batch number, purchase information, your reference, and reason for return. Please contact your local distributor office regarding product return/exchange.
DISPOSAL OF THE USED DEVICE
• Disposal of the used product should be done according to hospital procedures, State Regulations, national legislation or regional legislation.
• Device should be disposed after use on individual patient during a single procedure.
• Disposal of the packaging material should be done as biomedical waste.
• Any open packaging should be disposed as biomedical waste.
• Leftover products should be disposed as biomedical waste
REPORTING
In case of any adverse events and/or potentially sight-threatening complications that may reasonably be regarded as product-related and that were not previously expected in nature, severity or occurrence, contact the manufacturer or authorized representative and competent authority of the member state where the user and/or patient is established.