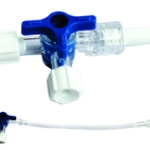
Description
Accu Line (Pressure Monitoring Line) is a precision medical tubing set designed for accurate and reliable pressure monitoring in clinical settings. It enables stable transmission of pressure signals between the patient and monitoring equipment, ensuring consistent performance in invasive and non-invasive procedures. Made from high-quality medical-grade materials, it offers kink resistance, secure connectivity, and clear pressure waveform transmission to support informed clinical decisions in critical care and operating environments.
Please Download eIFU
DESCRIPTION:
The SPM’s Accu-Line is a sterile, single-use, Pyrogen-free Extension line. The Extension line is
available in different lengths ranging from 10cm to 250cm PVC Tube. The Extension Line consists of
male luer on both ends. The device is ethylene oxide sterilized.
INTENDED PURPOSE:
Extension Line is used to extend the infusion line during administration of IV fluid. It also provides a
pressure monitoring aid.
INDICATIONS FOR USE:
• SPM’s Extension Line is intended use to connection and extension infusion or transfusion
sets for administration liquids or blood into the circulation system by using of intravenous
catheter and cannula.
• Allow pressure monitoring.
INTENDED PATIENT POPULATIONS:
Accu-Line Extension line is intended to be used in patients irrespective of age and gender.
INTENDED USERS:
Accu-Line Extension line is intended to be used by the trained healthcare professionals including
doctors, registered practitioners, and paramedics or nursing staff.
CLINICAL BENEFITS:
• The transparent, smooth and kink-resistant PVC tube (latex-free) allows seamless
connection
• 6% taper in luer lock allows connection with a variety of medical devices.
PRINCIPLE OF OPERATION:
The Extension Line allows the extension of the vascular access device like infusion set, cannula or
port, etc. It offers more flexibility and convenience during treatments. It also allows pressure
monitoring.
PERFORMANCE CHARACTERSTICS:
• Interoperability with other vascular access devices like IV cannula or infusion set.
• The extension line allows pressure monitoring.
MATERIAL USED:
• Extension Tube – PVC
• Female Ports – Poly Propylene
• Luer Connector- HDPE
• Luer protecting cap- Poly Propylene
POTENTIAL COMPLICATIONS:
Fluid leakage and cross-contamination, Difficulty turning, Incompatibility with medications, Blockage
and obstruction, sterility breakdown, misalignment of ports, Material deterioration, and limited
durability in high-pressure.
INSTRUCTIONS FOR USE:
• Step 1. Peel Open the pouch.
• Step 2. Unscrew the luer cap of three way stop cock female luer.
• Step 3. Connect male portion of I.V. Set with female luer of Three Way Stop cock.
• Step 4. Remove luer cap of male luer tubing.
• Step 5. Hold male luer of tubing upright and open clamp of I.V. Set and allow the
solution to displace air entirely from tubing.
• Step 6. Close clamp.
• Step 7. Connect male luer of tubing to venepuncture device.
HOW SUPPLIED
The Extension Line is supplied in sterile, single-use Paper pouch packaging, complete with
instructions for use and necessary accessories (if any).
WARNINGS
• For Professional Use Only: This device must be used only by trained healthcare
professionals
• Sterile Packaging: Do not use if the packaging is damaged or opened prior to use.
• Single Use Only: The Extension line is designed for single use. Do not reuse or re-sterilize.
• Proper Handling: Handle with care to avoid damage to the device, which could
compromise its sterility and functionality
• Allergic Reactions: Check patient history for allergies to materials in the Extension line.
• Inspect Before Use: Ensure all components are present and undamaged before use.
• Follow Instructions: Carefully follow the included instructions for use to avoid
complications
• Dispose Properly: Dispose of the device in accordance with local regulations for medical
waste
PRECAUTIONS:
• Check the integrity and functionality of the Extension Line before use. Do not use if the
package is open or damaged.
• Determine patient’s condition and vitals status during device application / Operation.
• When handling the product care should be taken to avoid damage from handling. Avoid
crushing and crimping damage to the product due to application of surgical instruments
such as forceps, etc.
• Conduct procedure under strict surgical protocol and ensure complete asepsis.
• Do not Re-sterilize. Do not Re-use.
• Single use only.
• Do use after the Expiry Date
CAUTIONS:
• Always check for leaks after changing the connection.
• Store in a cool and dry place
• Discard after single uses
• Do not use if the package is open or damage
• Do not store at extreme temperature and humidity
STORAGE:
Store at a controlled temperature (10-40°C). Do not store at freezing temperature.
EXPIRATION PERIOD:
The device has a shelf life of 5 years from the date of manufacturing. The product should not be
used after the indicated sterility expiration date.
RETURN OF DAMAGED PRODUCT:
Return the product in its original packing identified by the batch number, purchase information,
your reference and reason for return. Please contact your local distributor office regarding product
return/exchange.
DISPOSAL OF THE USED DEVICE:
• Disposal of the used product should be done according to hospital procedures, State
Regulations, National legislation or regional legislation.
• Device should be disposed after use on individual patient during a single procedure.
• Disposal of the packaging material should be done as biomedical waste.
• Any open packaging should be done as biomedical waste.
• Leftover products should be disposed as biomedical waste.
REPORTING:
In case of any adverse events and/or potential threat/ complications that may reasonably by
regarded as product related and that were not previously expected in nature, severity or occurrence,
contact the manufacturer or authorized representative and competent authority of the member
state where the user and/or patient is established.