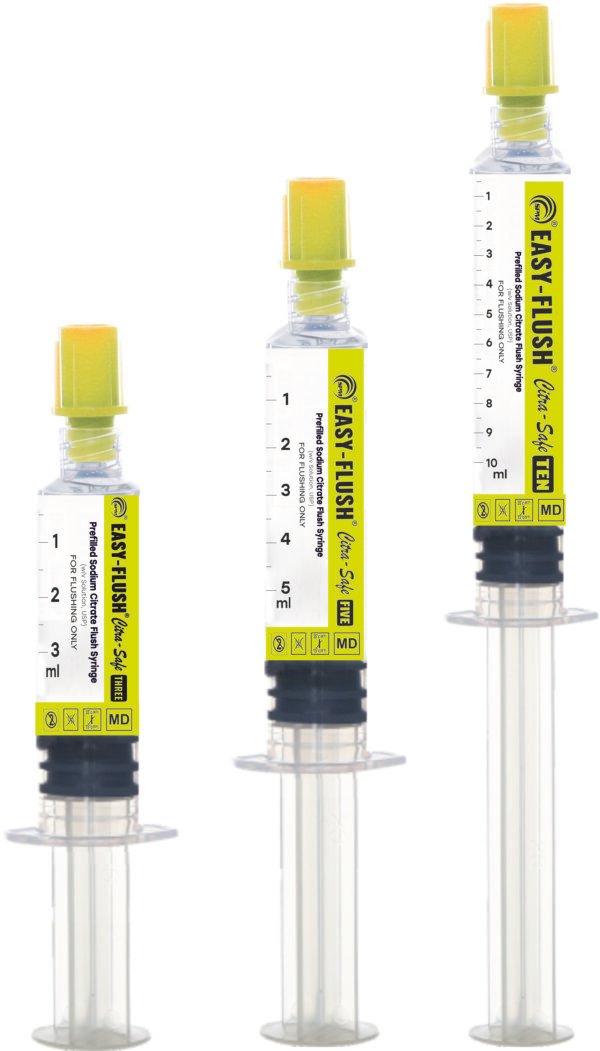
Description
Available in
• Sizes: 3ml, 5ml, 70ml
• Packaging: Individual Blister Pack
50ml1BO7RZS40/32030/240
| Brand | Size | Product code | Qty. (Inner / Outer)/ Europe |
Qty. (Inner / Outer)/ USA |
|---|---|---|---|---|
| Prefilled Sodium Citrate
Flush syringe |
3ml | 1DQ783A | 50/400 | 50/400 |
| 5ml | 1DQ7B5A | 50/400 | 50/400 | |
| 10ml | 1DQ7BXA | 40/320 | 40/320 |
Please Download eIFU
DESCRIPTION:
The SPM’S Prefilled Sodium Citrate Flush Syringe is a standard polypropylene luer lock syringe pre-filled
with sterile 4% w/v Sodium Citrate solution. The syringe facilitates safe and quick flushing of venous
access device using Sodium Citrate solution. The Prefilled Sodium Citrate Flush Syringe is non-pyrogenic,
single use, non-toxic device and free from natural rubber latex and DEHP. The pre-filled syringe is sterilized
by Gamma irradiation sterilization. The duration of the use is Short term.
A Sodium citrate solution is used to prevent vascular access systems from becoming blocked and to help
remove any medication that may be left at the catheter site designed for manual use.
INTENDED PURPOSE:
Prefilled Syringe with Sodium Citrate solution is intended for flushing compatible intravenous
administration sets and indwelling intravenous access devices.
INDICATIONS FOR USE:
• Prefilled syringe with Sodium Citrate solution is intended to maintain patency of an indwelling
intravenous access device.
• Prefilled syringe with Sodium Citrate solution is preventing clotting in IV access medical devices (e.g.,
catheters) before and during operation/procedure.
INTENDED PATIENT POPULATION:
Prefilled Syringe with Sodium Citrate solution is intended to be used in patients who required venous
access such as haemodialysis, CVC, etc.
INTENDED USER:
The intended users for the Prefilled Syringe with Sodium Citrate solution are healthcare professionals like
medical practitioner, nurses, and other paramedic staff.
INTENDED USE ENVIRONMENT:
Prefilled Syringe with Sodium Citrate solution is intended to be used in healthcare environment like
hospitals and clinics.
POTENTIAL COMPLICATIONS:
Possible complications or adverse reactions associated with Prefilled Syringe with Sodium Citrate solution,
may include haemorrhage, thrombocytopenia, injection site irritation, general hypersensitivity reactions,
etc. The chelation of calcium ions may resulting in hypocalcaemia and hypernatremia, as well as possible
emergence of paraesthesia and/or dysgeusia
INSTRUCTIONS FOR USE:
The Prefilled Syringe with Sodium Citrate solution, is a sterile, pyrogen-free device. If the tip cap is in place,
the syringe is intact & there is no evidence of leakage. Handle aseptically.
• Step 1. Carefully inspect the package. Do not use, if the package is open or syringe is damaged.
(Figure.1)
• Step 2. Remove plastic packaging by tearing along the perforation (Ribbon) or Peel open the pouch
(Blister). (Figure.2)
• Step 3. Carefully inspect if a solution is discoloured, cloudy, hazy, or contains a precipitate, or if the
syringe is damaged. (Figure.3)
• Step 4. Clear the catheter of any remaining closure solutions by aspirating into an empty sterile syringe
(Figure.4)
• Step 5. Flush each catheter lumens using sterile isotonic solution (0.9% Sodium Chloride) to remove
any remaining blood components from previous procedures.
• Step 6. With the tip cap is still on, Push plunger rod slightly to activate the syringe. Never draw back
the plunger rod as the product may contaminate.
• Step 7. Remove the tip cap by twisting it off using aseptic technique. Hold the syringe unit upright and
prime it to expel any air bubbles (if present). (Figure.5)
• Step 8. The syringe is now ready to use. As per institutional protocol, attach the flush syringe to the
access device without any contamination. (Figure.6)
• Step 9. Slowly inject the required amount of pre-filled solution by slowly pressing the plunger to avoid
excessive pressure. If you experience plunger resistance, it is recommended, do not apply excessive
force. Do not pull or bend the plunger. (Figure.7)
• Step 10. Use in accordance with intravenous tubing or indwelling device manufacturer’s
recommendation. After flushing, gently remove the syringe from the access device. (Figure.8)
• Step 11. Discard the empty unit after use. Discard any unused portion. Do not reuse disposable
syringes. (Figure.9)
HOW SUPPLIED:
The Prefilled Syringe with Sodium Citrate solution is supplied in both sterile fluid path and externally
sterile, single-use, packed individually in Ribbon or Paper pouch, complete with instructions for use and
necessary accessories (if any).
WARNINGS:
• For Professional Use Only: This device must be used by trained healthcare professionals only.
• Sterile Packaging: Do not use if the packaging is damaged or opened prior to use.
• Not Intended for Intramuscular use: The Prefilled Syringe with Sodium Citrate solution, is not
intended for Intramuscular use.
• Discontinue its use: Discontinue use of the syringe, if coagulation test is excessively prolonged, or if
haemorrhage or thrombocytopenia occurs.
• Single Use Only: The Sodium Citrate pre-filled flush syringe is designed for single use. The Reprocessing of the device may compromise the sterility, biocompatibility, and functional integrity of
the device.
• Proper Handling: Handle with care to avoid damage to the device, which could compromise its
sterility and functionality.
• Allergic Reactions: Check patient history for allergies to materials used in the Sodium Citrate
prefilled syringe.
• Inspect Before Use: Ensure all components are present and undamaged before use. Do not use if
the prefilled solution appears cloudy, coloured, or contains any precipitate of sended particulate
matter.
• Follow Instructions: Carefully follow the included instructions for use to avoid complications.
• Dispose Properly: Dispose of the device in accordance with local regulations for medical waste.
PRECAUTIONS:
• The Prefilled Syringe with Sodium Citrate solution, is designed to be used for direct flushing or
with ISO luer compliant components for Intravenous application.
• Perform patient’s Blood coagulation tests before the Sodium Citrate flushing.
• Graduation is provided on the syringe, to inject the required dose.
• Check the product for defects/cracks or change in appearance of pre-filled solution.
• Avoid crushing and crimping damage to the product due to the application of surgical instruments
like forceps, etc.
CAUTIONS:
• The Federal Law restricts this device to sale by for or order by physician.
• Carefully read all instructions prior to use.
• Hold the syringe in upward direction and expel the bubbles from the syringe.
• For single use only. Discard after single use.
• Don’t use if the package is open or damage.
• Do not store at extreme temperature and humidity.
• Dispose the contaminated/ used product as per local applicable laws.
STORAGE:
Store in cool and dry place at controlled temperature (5 to 30°C or 41 to 86°F). Do not freeze. Store away
from direct sunlight.
EXPIRATION PERIOD:
The Prefilled Syringe with Sodium Citrate 4% (w/v) solution has a shelf-life of 36 months (3 years) from
the date of manufacturing. The product should not be used after the indicated sterility expiration date.
RETURN OF DAMAGED PRODUCT:
Return the product in its original packing identified by the batch number, purchase information, your
reference and reason for return. Please contact your local distributor office regarding product
return/exchange.
DISPOSAL OF THE USED DEVICE:
• Disposal of the used product should be done according to hospital procedures, regional legislation
or National legislation.
• Device should be disposed after use on individual patient during a single procedure.
• Disposal of the packaging material should be done as biomedical waste.
• Any open packaging should be done as biomedical waste.
• Leftover products should be disposed as biomedical waste.
REPORTING:
In case of any adverse events and/or potentially sight threatening complications that may reasonably by
regarded as product related and that were not previously expected in nature, severity or occurrence,
contact the manufacturer or authorized representative and competent authority of the member state
where the user and/or patient is established.