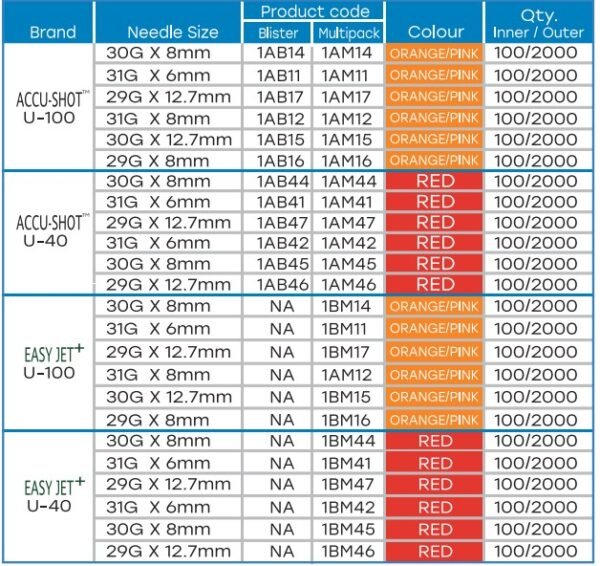
Description
Available also
- 29G, 30G, 31G & Needle sizes 6mm, 8mm, & 12.7mm
- Packaging: Individual pack in Blister, Ribbon, & 10pcs. in Multipack.
Please Download eIFU
DESCRIPTION
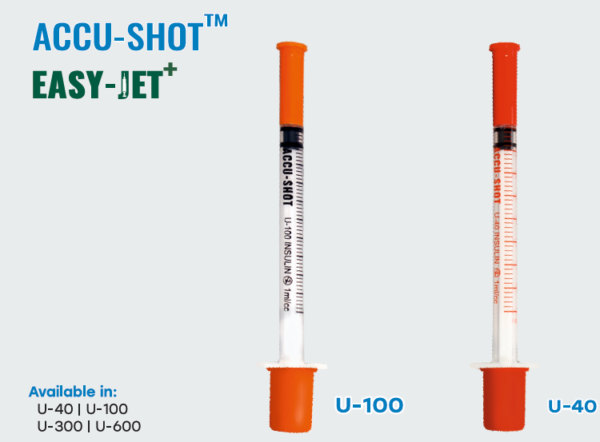
The SPM’s Accu-Shot™ and Easy-Jet+ Insulin Syringe is a plastic syringe designed for
subcutaneous injection of a desired dose of insulin. The insulin syringe consists of a
graduated barrel, plunger rod and needle/hub assembly. The insulin syringes are
sterile, single use, and non-pyrogenic. This device operates on the same principle as
piston syringes. It is sterilized by ethylene oxide (EO).
INTENDED USE (Purpose)
The Insulin Syringe is intended for the safe administration of insulin in
insulin‑dependent diabetic patients.
INDICATIONS FOR USE
The Insulin Syringe is designed for the safe administration of insulin in diabetic
patients.
INTENDED PATIENT POPULATION
The Insulin Syringe is intended for diabetic patients irrespective of age and gender.
INTENDED USER
Professional medical staff including physicians, nurses, therapists, and paramedic staff.
(Also suitable for trained patients/self‑administration as per institutional or physician
guidance.)
INTENDED USE ENVIRONMENT
Home setting, hospital, or clinical setting.
STERILISATION
The device is supplied sterile and non‑pyrogenic, sterilised using ethylene oxide (EO).
CLINICAL BENEFITS
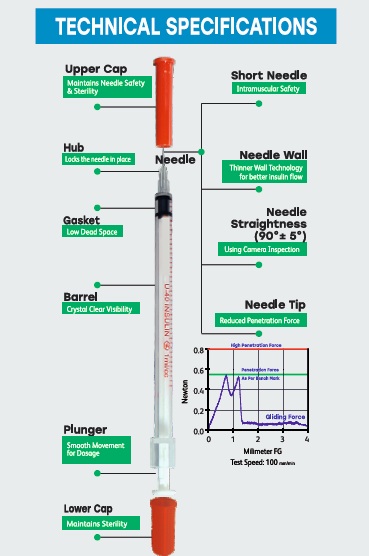
Short and thin attached needle for more safety and comfort.
Low dead space for more accurate dosage and minimal wastage.
Bold and clear graduation markings for ease of reading.
PRINCIPLE OF OPERATION
The SPM’s Accu-Shot™ and Easy-Jet+ syringe is used for the administration of insulin via
a fine needle. It consists of a marked barrel, plunger, and fine needle with a protective
needle cap. The user draws insulin into the syringe, removes air bubbles, and injects it
into the fatty tissue under the skin to enable absorption into the bloodstream and
regulation of blood glucose levels.
PERFORMANCE CHARACTERISTICS
Provides safe and accurate insulin administration for diabetic patients.
Low dead space design for accurate dosing and minimal drug wastage.
Available in red and orange colour options.
Bottom cap and needle cap enhance handling and safety.
DEVICE COMPONENT DESCRIPTION (Materials)
Barrel – Polypropylene (PP)
Plunger – Polypropylene (PP)
Gasket – Thermoplastic Elastomer (TPE)
Needle Cap – Polyethylene (PE)
Plunger (Bottom) Cap – Polyethylene (PE)
Lubricant – Silicone oil
Adhesive – UV glue
Needle/Hub Assembly – Stainless steel cannula with integral hub
COMPONENTS OF THE DEVICE
Barrel (graduated)
Plunger rod
Gasket
Needle with hub (attached)
Needle cap
Plunger bottom cap
POTENTIAL COMPLICATIONS
Hypoglycaemia due to overdosing or incorrect administration.
Local pain, bruising, or bleeding at injection site.
Infection or abscess if asepsis is not maintained.
Lipohypertrophy/lipoatrophy with repeated injections at the same site.
Allergic reactions (to materials or insulin).
Needle‑stick injury to user or healthcare professional.
Cross‑contamination with reuse.
DIRECTIONS FOR USE
• Verify packaging: Use only if the sterile pack is intact and within expiry.
• Prepare dose: Confirm the insulin concentration (U‑40 or U‑100) matches the
syringe type. Wash hands.
HOW SUPPLIED
The SPM’s Insulin Syringe is supplied sterile, single‑use, in Blister / Ribbon / Multi‑pack
pouch packaging, complete with instructions for use.
WARNINGS
Improper transport/handling may damage the device or packaging.
Product is non‑toxic, sterile & non‑pyrogenic if the package is unopened and
undamaged.
Single use only. Do not re‑sterilise, reuse, or re‑process.
Re‑sterilising can compromise performance and increase cross‑contamination risk.
Reuse may cause cross‑infection.
Remove the needle cap carefully to avoid needle‑stick injury.
Do not inject air into the patient; avoid manipulating the plunger in a way that affects
dose accuracy.
PRECAUTIONS
Read instructions before use.
Store in a clean, dry place; avoid direct sunlight; keep at room temperature.
Do not use after the expiry date.
Do not use if sterile packaging is damaged/opened prior to use.
Use immediately after opening.
CAUTIONS
Store in a cool, dry place.
Discard after single use.
Do not use if the package is open or damaged.
Do not store at extreme temperature or humidity.
Dispose of blood/medicine‑contaminated product per local regulations.
ADVERSE EVENTS
Phlebitis (not typical for s.c. injection; generally not expected)
Infection / toxic reaction
Injury / needle‑stick injury
Incomplete dose administration
Blood‑glucose instability (e.g., hypoglycaemia)
Bruising/haematoma at injection site
Lipodystrophy with poor site rotation
Allergic reactions
Cannulation failure (not applicable; syringe only)
STORAGE CONDITION
Store at a controlled temperature (10–40°C) in a cool, dry, dark place.
Note: Avoid exposure to direct sunlight. Exceeding the recommended storage
conditions may affect product quality.
SHELF LIFE
5 years from the date of manufacture. Do not use after the indicated sterility expiry
date.
RETURN OF DAMAGED PRODUCT
Return the product in its original packing identified by the batch number, purchase
information, your reference and reason for return. Please contact your local distributor
office regarding product return/exchange.
DISPOSAL
After use, this product may be a potential biohazard.
Dispose of after single‑patient use during a single procedure.
Dispose of the used product and packaging as biomedical waste according to hospital
procedures and local regulations.
REPORTING
In case of any adverse events and/or potential complications that may reasonably be
regarded as product‑related and that were not previously expected in nature, severity,
or occurrence, contact the manufacturer or authorised representative. Additionally,
notify the competent authority of the member state where the user and/or patient is
established, as per local regulatory requirements.