Description
Please Download eIFU – Click Here
DESCRIPTION:
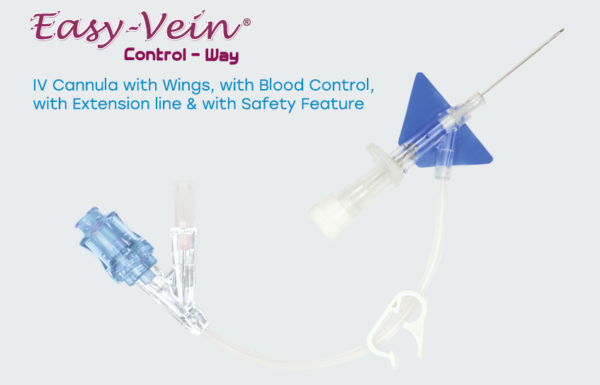
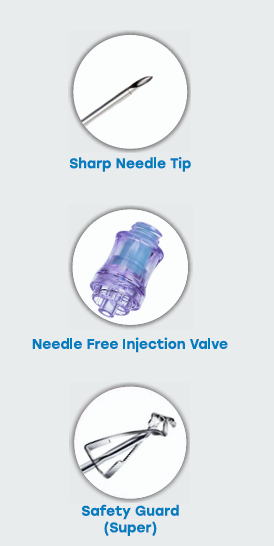
IV Cannula is a smooth, fine-tipped, tapered, radio- opaque tube with a three-faced sharp and
dual cutting- edge needle for cannulation. It is designed to introduce fluids, medications, and
blood components into the body via Vascular Access. It consists of a flashback
chamber for ensuring proper venous penetration, wings for fixing it to the penetration site, and
colour-coded injection ports for intermittent fluids and medication or blood sampling. It has a
self-activating safety mechanism. The safety guard automatically covers the needle tip after the
needle is withdrawn to minimise the risk of needle-stick injury and blood-borne infections
during needle withdrawal and disposal.
IV Cannula incorporating a closed system that minimizes blood backflow and reduces the risk
of exposure to healthcare personnel. The device includes an integrated extension tube for
remote handling, and a Luer lock interface to ensure secure, leak-proof connections with
infusion accessories.
INTENDED USE:
The IV Cannula is a sterile, single use medical device intended to provide peripheral vascular
access for short term use.
INDICATIONS FOR USE:
• Phlebotomy (blood sampling)
• Infusion of IV solution
• Provide nutritional support
• Maintain hydration and/or correct dehydration (if the patient is unable to take sufficient
oral fluids).
INTENDED USER:
The device is restricted for professional use only, i.e. Physicians, nurses and Paramedics trained
in handling the device.
INTENDED USE ENVIRONMENT:
Hospitals or clinics.
INTENDED PATIENT POPULATION:
IV Cannula is intended for patients irrespective of age and gender.
STERILISATION:
The device is supplied sterile and pyrogen-free, sterilized by ethylene oxide.
SHELF LIFE:
The device has a 5-year shelf life
COMPONENTS OF THE DEVICE:
• Wing body
• Hub cover
• Needle hub
• Teflon holder
• Port cap
• Luer lock
• Needle cover
• Silicon tube
• Needle
• Catheter
DEVICE COMPONENT DESCRIPTION:
• Stainless Steel AISI 304: Needle;
• Polyurethane Catheter
• Polypropylene (PP): Wing body, Needle cover, Needle Hub, Flash Back Chamber, Threaded
Stopper;
• Poly Acetel: Catheter Holder;
• Hight Density Polyethylene (HDPE): Port Cap;
• Silicon rubber: Silicon Tube
PRINCIPLE OF OPERATION:
IV cannula acts as a medium for the passage of fluid from gravity feed Infusion sets. The end
with luer connection connects with the infusion line and the end with cannula inserts into the
circulatory system.
CONTRAINDICATIONS:
• Not to be used in persons hypersensitive to any raw material used
• Infuse irritant fluids or drugs
• Delivery of viscous fluid
• Near sites of infection, fractured bones, oedema, and areas of surgery.
• Major arteries and veins,small veins, and previously cannulated veins
CLINICAL BENEFITS:
- Reduce the need for repeated needle prick
- Prevents accidental needle–stick injury
- Multiple delivery of medication or fluids
WARNING AND PRECAUTIONS:
• The device is intended for single use only. Do not reuse the device.
• Do not re-sterilize the device.
• Do not use the device if the sterile packaging is compromised, damaged or opened before
use.
• Do not use device after shelf life expires.
ADVERSE EVENTS:
• Phlebitis
• Thrombophlebitis
• Infection/ Toxic reaction
• Injury
• Incomplete infusion
• Blood stream infection
• Trauma to vessel
• Occlusion
• Needle stick injury
• Catheter migration
• Cannulation failure
POTENTIAL COMPLICATIONS:
Possible complications with IV Cannula with wings, extension line, and safety features
valve may encounter some challenges like:
• Use of contaminated cannula may result in transmission of infections.
• The improper handling may result in leakage or injury.
• Air entering the bloodstream if not properly primed or if there is a leak.
STORAGE CONDITION:
Store at a controlled temperature between 10°C and 40°C at cool, dry and dark places.
Note: Avoid exposure to direct sunlight. Exceeding the maximum recommended storage
temperature may lead to deterioration of the IV Cannula Quality.
INSTRUCTIONS FOR USE:
1. Carefully select and aseptically prepare the site.
2. Select suitable size of cannula and inspect visually to ensure that package is intact. Do not
use if the package is damaged or previously or previously opened. Check for visible damage or
contamination.
3. Open the sterile pack from the peel side clearly marked as “PEEL OPEN.” Use aseptic
technique while opening.
4. Remove cannula from sterile packaging without touching the catheter part.
5. Grip the cannula from the injection port & projection provided on the port (For I.V.
Cannula having injection port).
6. Ensure Vent Plug is secure & make sure clamp is not engaged. (for Closed I.V. Cannula)
7. For 26G cannula, especially in children and elderly patients, flush the extension line with
sterile saline prior to use to confirm patency and minimize flow resistance. (for Closed I.V
Cannula)
8. Grip the cannula so that the needle and catheter are properly aligned (For I.V. Cannula not
having injection port).
9. Remove the protective needle cover just before insertion.
10. Perform venipuncture & check for flash back of blood in flashback chamber (22G to 14G)
or along the catheter length (24G to 26G). Once flashback is visible, lower the angle of the
device slightly and advance the catheter.
11. Do not retract the needle before the catheter is fully advanced. Advance the catheter into
vein and simultaneously withdraw the needle.
12. Immediately and completely withdraw the needle after catheter advancement to prevent
clot formation. Press the vein just after the catheter tip and discard the needle in an
appropriate sharp’s container.
13. Secure the cannula to the patient with sterile adhesive dressing to prevent dislodgement.
14. For closed IV cannula, ensure the vent plug is securely in place before use.
15. Connect to the I.V. Infusion set line.
16. Cover the puncture site with sterile dressing.
17. Drugs can be injected with the help of syringe without needle through integrated injection
port after removing the port cap. Close the injection port after use (for I.V. Cannula having
injection port).
18. Perform routine monitoring & venipuncture site maintenance according to medical
norms.
19. Document the insertion and removal time, and monitor the site for signs of infection,
infiltration, or phlebitis.
20. Do not reuse or resterilise the device under any circumstances. This is a single-use device
only.
RETURN OF DAMAGED PRODUCT:
Return the product in its original packing identified by the batch number, purchase
information, your reference and reason for return. Please contact your local distributor office
regarding product return/exchange.
REPORTING:
In case of any adverse events and/or potential threat/ complications that may reasonably by
regarded as product related and that were not previously expected in nature, severity or
occurrence, contact the manufacturer or authorized representative and competent authority
of the member state where the user and/or patient is established.
DISPOSAL:
• After use, this product may be a potential biohazard.
• Device should be disposed of after use on individual patients during a single procedure.
• The used product should be disposed of as biomedical waste according to hospital
procedures and local waste disposal regulations.